Yomogi Saccharomyces cerevisiae (boulardii) Probiotic
Price range: $16.80 through $32.80
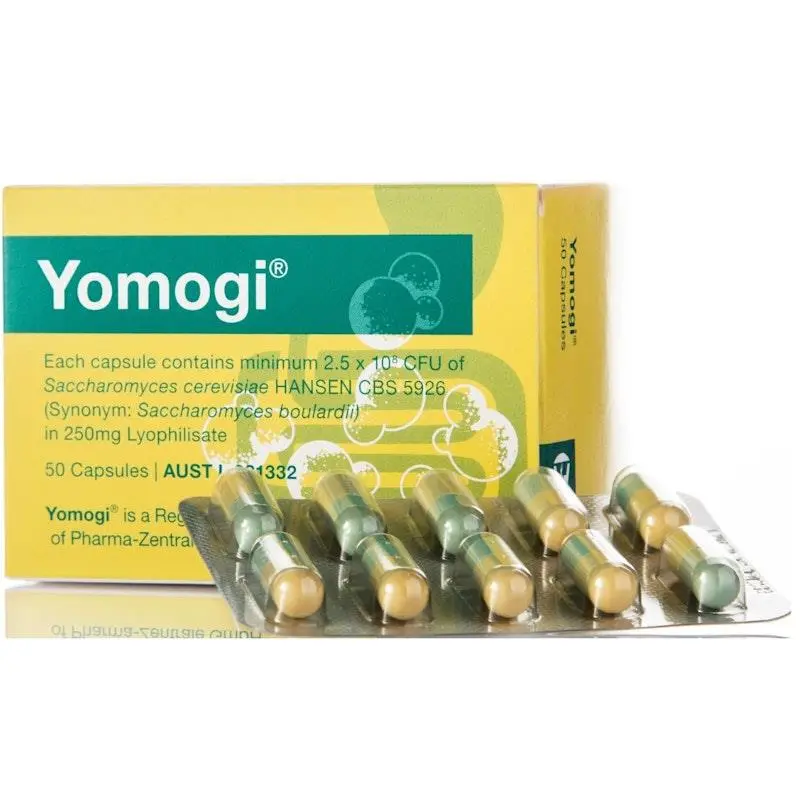
Probiotic supplement: Saccharomyces cerevisiae (boulardii) HANSEN CBS 5926 yeast strain.
Yomogi heat-stable, pharmaceutical-grade formulation requiring no refrigeration, ideal for travel and antibiotic support.
What is Yomogi and who is it for?
Yomogi is a pharmaceutical-grade probiotic containing Saccharomyces cerevisiae (boulardii), a tropical yeast strain with a documented history dating back to the 1920s. Unlike bacterial probiotics that colonise your gut, S. boulardii is a transient yeast that passes through your digestive system while exerting its beneficial effects. This makes it particularly useful during periods when your gut microbiota needs temporary support, especially during or after antibiotic use.
This product makes sense if you’re managing antibiotic-associated diarrhoea, preparing for travel to regions where traveller’s diarrhoea is common, or looking for a heat-stable probiotic that doesn’t require refrigeration. The lyophilised formulation and blister-sealed packaging make it genuinely practical for travel, unlike many probiotics that need careful temperature management.
Fairfield Nutrition Thoughts
Antibiotic-associated diarrhoea (AAD) is one of the most common adverse effects of antibiotic treatment, occurring in 10-30% of patients depending on the antibiotic class. Saccharomyces boulardii (Yomogi) addresses this specific problem as it’s a transient organism that exerts effects as it passes through rather than establishing permanent residence.
The clinical evidence for S. boulardii in AAD is substantial and well-documented, with multiple randomised controlled trials demonstrating reduction in both frequency and severity of diarrhoea. What makes Yomogi’s formulation compelling is the combination of strain specificity (HANSEN CBS 5926 / CNCM I-745, the most researched strain), heat stability eliminating cold-chain dependency, and pharmaceutical-grade blister packaging that maintains viability without refrigeration. This is particularly relevant for Australian consumers managing travel or those in warmer climates where refrigerated probiotics become impractical. It may also prevent the likelihood of travellers’ diarrhoea.
Why This Formulation / How It Works
Yomogi differs from multi-strain bacterial probiotics in a fundamental way: it’s a single-strain yeast designed for temporary support rather than long-term colonisation. The HANSEN CBS 5926 strain is the most extensively researched S. boulardii strain globally, with decades of clinical use and documented mechanisms. This formulation eliminates the need for refrigeration while maintaining minimum 2.5 billion CFU per capsule at batch release, making it genuinely stable at room temperature and suitable for travel without compromising potency.
S. boulardii functions as a transient organism in your digestive tract, meaning it passes through rather than establishing permanent residence. As it moves through your intestines, it exerts several beneficial effects: it produces compounds that support intestinal barrier function, provides competition for pathogenic organisms, and produces short-chain fatty acids that support healthy colonic tissue. This transient nature is actually advantageous during antibiotic use, because the yeast won’t be affected by the antibiotic in the way that beneficial bacteria would be.
Support during antibiotic use: When antibiotics disrupt your gut microbiota, the resulting dysbiosis creates an environment where pathogenic organisms like Clostridioides difficile can establish themselves. S. boulardii reduces this risk by maintaining microbial diversity and supporting intestinal barrier integrity while your beneficial bacterial species recover after antibiotic treatment ends.
Transient yeast mechanism: Unlike bacterial probiotics that attempt to colonise and establish permanent populations, S. boulardii exerts its effects during transit through your digestive system. This means it works effectively even in the presence of antibiotics and doesn’t require your gut to provide the conditions necessary for permanent colonisation.
When to Consider This
This formulation provides targeted support for specific gastrointestinal scenarios. Yomogi makes sense if you’re:
Scenario 1: Starting a course of antibiotics and wanting to reduce the risk of antibiotic-associated diarrhoea, which affects up to 30% of people taking certain antibiotic classes.
Scenario 2: Travelling to regions where traveller’s diarrhoea is common and want a heat-stable probiotic that doesn’t require cold storage or refrigeration during your trip.
Scenario 3: Recovering from recent antibiotic use and experiencing ongoing digestive dysbiosis, as S. boulardii supports beneficial microbiota recovery without being affected by residual antibiotic presence.
Supplement strategy: S. boulardii is typically introduced at the start of antibiotic treatment or 5 days before travel, rather than as ongoing daily support. Many practitioners recommend using it for the duration of antibiotic therapy plus 2 weeks after completion, then transitioning to multi-strain bacterial probiotics once your microbiota has begun recovering. It’s often used alongside dietary support (adequate fibre, fermented foods) and sometimes with prebiotic compounds, though Yomogi can be used independently.
⚠️ Not suitable for: Immunocompromised or hospitalised patients, patients with central venous catheters, hypersensitivity to Saccharomyces cerevisiae or yeast, pregnancy and breastfeeding, lactose intolerance
How to Use
Standard dosage: Adults and children over 6 years take 1 capsule once or twice daily. For children aged 2-6 years, open the capsule and stir the contents into food or liquid.
For traveller’s diarrhoea prevention: Take 1 capsule once or twice daily starting 5 days before departure and continue for the duration of travel.
During antibiotic use: Take 1 capsule once or twice daily. Many practitioners recommend starting this when you begin your antibiotic course and continuing for 2 weeks after completing antibiotics.
Yomogi requires no refrigeration due to its lyophilised formulation, making it genuinely convenient for travel or storage in warm environments. Each capsule contains a minimum of 2.5 billion CFU at batch release. Always follow your healthcare professional’s specific recommendations, particularly if you have any pre-existing conditions or are taking other medications.
Ingredients
Per capsule:
Saccharomyces cerevisiae (boulardii) HANSEN CBS 5926 / CNCM I-745: 250 mg (minimum 2.5 billion CFU at batch release)
Other ingredients: Capsule (hypromellose), microcrystalline cellulose
Allergen information: Contains lactose. Free from common allergens including gluten, soy, and dairy (aside from lactose content).
Common Questions
How is S. boulardii different from the multi-strain probiotics I’ve seen?
S. boulardii is a yeast, not a bacterium, and it’s transient, meaning it passes through your gut rather than establishing permanent colonies. This makes it particularly useful during antibiotic treatment, when you want temporary support without organisms that might be killed by the antibiotic. It’s designed for specific scenarios (antibiotic use, traveller’s diarrhoea, with anti-microbials) although it’s safet allows it to be used for general daily microbiota maintenance.
Can I take this with my antibiotics?
Yes, that’s actually when it’s most useful. Unlike bacterial probiotics that antibiotics might suppress, S. boulardii works effectively during antibiotic treatment because it’s not affected by the antibiotic. Many practitioners recommend starting it when you begin your antibiotic course and continuing for 2 weeks after completion.
Why doesn’t this need refrigeration like other probiotics?
Yomogi uses lyophilisation (freeze-drying) technology to stabilise the yeast cells, making them viable at room temperature. This is a significant practical advantage for travel or warm climates, and it means the product maintains its potency without cold-chain dependency.
How long should I take this for?
This depends on your situation. For antibiotic-associated diarrhoea, most people use it during the antibiotic course plus 2 weeks after. For traveller’s diarrhoea prevention, start 5 days before travel and continue during your trip. It’s not typically used as indefinite daily support like multi-strain probiotics; it’s more targeted to specific situations.
Is this suitable for children?
Children over 6 years can take 1 capsule once or twice daily as directed. Children aged 2-6 years can use it if the capsule is opened and contents stirred into food or liquid. Children under 2 years and infants should not use this product.
Warnings & Storage
Do not use if: Blister backing is damaged or broken, you are hypersensitive or allergic to Saccharomyces cerevisiae or yeast, you are immunocompromised or hospitalised with serious illness, you have a central venous catheter, you are pregnant or breastfeeding, you have lactose intolerance.
Cautions: If symptoms persist, talk to your healthcare professional. Seek medical advice if diarrhoea persists for more than 6 hours in infants under 6 months, 12 hours in children under 3 years, 24 hours in children aged 3-6 years, or 48 hours in adults and children over 6 years. Contains lactose.
Storage: Store in a cool, dry place away from direct sunlight. No refrigeration required.
Always consult your healthcare professional before use if you have any pre-existing conditions or are taking medications.
| Weight | 0.041 kg |
|---|---|
| Dimensions | 6.6 × 6.2 × 10.6 cm |
| Size | 20 caps, 50 caps |